|
11/10/2023 0 Comments Atomic number of helium 
Some of the helium-3 found in the terrestrial atmosphere is also an artifact of atmospheric and underwater nuclear weapons testing. Helium-3 is also thought to be a natural nucleogenic and cosmogenic nuclide, one produced when lithium is bombarded by natural neutrons, which can be released by spontaneous fission and by nuclear reactions with cosmic rays. Helium-3 occurs as a primordial nuclide, escaping from Earth's crust into its atmosphere and into outer space over millions of years. 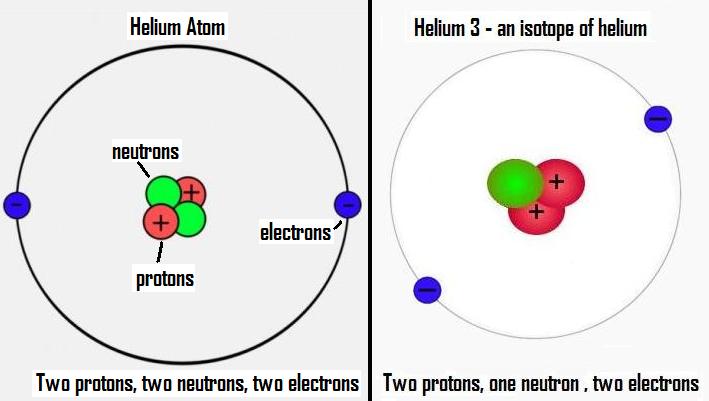
Other than protium (ordinary hydrogen), helium-3 is the only stable isotope of any element with more protons than neutrons. Helium-3 ( 3He see also helion) is a light, stable isotope of helium with two protons and one neutron (in contrast, the most common isotope, helium-4 has two protons and two neutrons). For the record label, see Helium 3 (record label). The name for the element comes from the Greek god of the Sun, Helios.This article is about the isotope. Evidence of helium's existence came from the observation of a yellow spectral line from the sun.Though helium is non-toxic, breathing it can result in asphyxiation due to oxygen deprivation. Although many people think inhaling helium makes a voice sound higher, it doesn't actually alter the pitch. Inhaling helium temporarily changes the sound of a person's voice.In other words, helium is found as single atoms of the element. 
Helium is monatomic under standard conditions.Helium is the second least reactive noble gas (after neon). It is considered the real gas that most closely approximates the behavior of an ideal gas.Most helium is obtained by extracting it from natural gas. Its uses include helium party balloons, as a protective inert atmosphere for chemistry storage and reactions, and for cooling superconducting magnets for NMR spectrometers and MRI machines.Under high pressure, disodium helide (HeNa 2), the clathrate-like titanate La 2/3-xLi 3xTiO 3He, the silicate crystobalite He II (SiO 2He), dihelium arsenolite (AsO 6♲He), and NeHe 2 may exist. In order to bond it to another element, it would need to be ionized or pressurized. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
